Ionic bonding in sodium chloride, artwork C017 / 7243
![]()

Wall Art and Photo Gifts from Science Photo Library
Ionic bonding in sodium chloride, artwork C017 / 7243
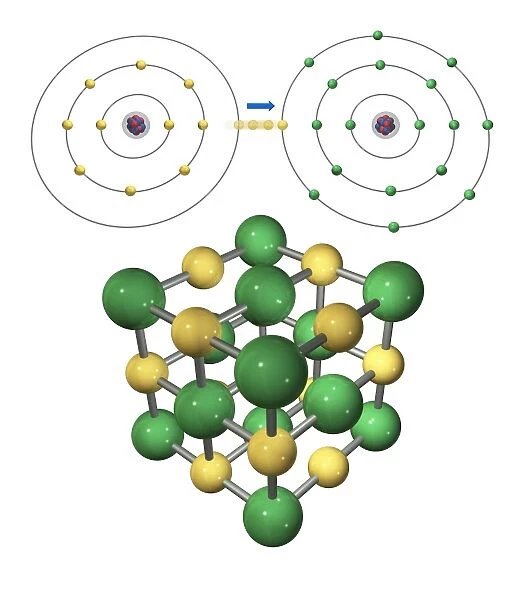
Ionic bonding in sodium chloride. Computer artwork showing the formation (top) of sodium chloride (common salt) and a model of the cubic crystal lattice (bottom) created by this chemical reaction. This is a classic case of ionic bonding, a type of chemical bond formed through an electrostatic attraction between two oppositely charged ions. Ionic bonds are formed due to the attraction between an atom that has lost one or more electron (known as a cation) and an atom that has gained one or more electrons (known as an anion). In the formation of sodium chloride, the sodium atom (top left) lost one electron, which is captured by the chlorine atom (top right). In the cubic crystal lattice, sodium ions (Na+) are represented by yellow spheres and chlorine ions
Science Photo Library features Science and Medical images including photos and illustrations
Media ID 9232907
© CARLOS CLARIVAN/SCIENCE PHOTO LIBRARY
Atom Atoms Attraction Ball And Stick Bond Bonds Charges Electrostatic Molecules Nacl Reaction Salt Sodium Chloride Cation Cutouts
EDITORS COMMENTS
This photo print, titled "Ionic bonding in sodium chloride" showcases the intricate artwork C017 / 7243. The image beautifully depicts the formation of sodium chloride, commonly known as salt, through a chemical reaction. At the top of the artwork, we witness this process taking place, while at the bottom, a model of the cubic crystal lattice created by this reaction is displayed. The artwork perfectly captures one of chemistry's classic cases: ionic bonding. This type of chemical bond occurs when two oppositely charged ions are attracted to each other due to their opposite charges. In sodium chloride's formation depicted here, we observe how an atom loses one electron (known as a cation) and another atom gains that electron (known as an anion). Specifically, the sodium atom on the top left surrenders its electron which is then captured by the chlorine atom on the top right. Within this cubic crystal lattice representation shown in yellow and white against a clean background, we can clearly see how these positively charged sodium ions (Na+) and negatively charged chlorine ions come together to form stable salt molecules. Carlos Clarivan from Science Photo Library has masterfully crafted this illustration with precision and attention to detail. Through his work, he highlights key elements such as structure, bonds formed through electrostatic attraction between atoms with opposite charges along with various cutouts representing different components involved in ionic bonding. Overall, this artwork serves as both educational material for students studying chemistry and a visually stunning piece for enthusiasts fascinated by science's wonders.
MADE IN THE USA
Safe Shipping with 30 Day Money Back Guarantee
FREE PERSONALISATION*
We are proud to offer a range of customisation features including Personalised Captions, Color Filters and Picture Zoom Tools
SECURE PAYMENTS
We happily accept a wide range of payment options so you can pay for the things you need in the way that is most convenient for you
* Options may vary by product and licensing agreement. Zoomed Pictures can be adjusted in the Cart.